Study design and study setting
A cross sectional study was conducted in outpatient department of ENT and Dental clinics at Aga Khan University Hospital (AKUH), recognized as the largest private tertiary care hospital in Karachi, Pakistan accredited by Joint Commission International (JCi) This facility provides comprehensive care to cancer patients serving diverse ethnic and socioeconomic backgrounds, and has benefitted more than 3 million cancer patients to date22. Additionally, AKUH actively promotes research activities and maintains a well-organized patient registry.
Study population, sampling technique and sample size
The participants in this study were patients who were 18 years and above, treated for HNC at Aga Khan University Hospital (AKUH). The study specifically included HNC survivors who were within the first year after completing their cancer treatment23, coming for their routine follow up at ENT and Dental clinics at AKUH. All the included patients had one of the three molars and a central incisor in upper and lower dentition at the time of follow up (these teeth were required to measure the Oral hygiene status using Oral Hygiene Index- simplified). We excluded patients who didn’t give their consent to participate in the study or had any debilitative conditions or cognitive disabilities confirmed by their medical reports or had trismus i.e. mouth opening < 20 mm (as it hindered the oral examination of the participant)22,24.
Participants were recruited using a non-probability purposive sampling technique. The study aimed to achieve its objectives with a minimum sample of 76 HNC-treated patients, considering an 80% statistical power and a significance level of 0.05. adjustments were made considering a 10% non-response rate, with an anticipated mean score 5 units higher than the hypothesized value of 16.7 in our population, as advised by the subject specialist19.
All patients treated for HNC at AKUH, between January 2021 and September 2022 were screened via their medical records. Patients who were within their first year of post-cancer treatment were contacted by phone to inquire about their upcoming follow-up visit to the ENT or Dental clinics at AKUH. On the scheduled follow-up day, we approached these patients to check for eligibility (i.e. for mouth opening and presence of at least one molar and incisor in each quadrant). Eligible patients were invited to participate in the study after explaining the purpose and scope of the study. Those who provided their written consent were interviewed and examined (Fig. 2). This study was approved by the Ethical review committee of Aga Khan University (2022-7178-21416).
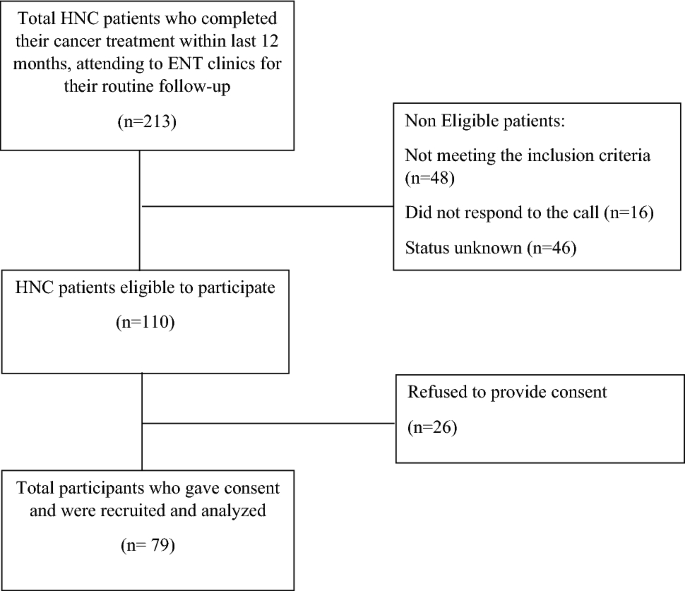
Flowchart of the process of study participant’s recruitment.
Variables and tools
Data was collected through a structured questionnaire, comprising sociodemographic variables, treatment related factors, oral hygiene practices and oral clinical parameters (oral mucositis and oral hygiene status).
Demographic and other variables
Sociodemographic variables included patients age recorded as (< 52 years’ vs ≥ 52 years)25, gender, marital status, BMI recorded as (underweight ≤ 18.5 kg/ m2, normal weight = ≥ 18.5 kg/ m2)26, level of education and monthly income. Treatment related factors included tumor stage at diagnosis according to AJCC stage system (stage I, stage II, stage III, stage IV), type of treatment received (without RT vs with RT), total dose of radiation received, elapsed time since treatment (< 6 months’ vs ≥ 6 months), number of follow up visits and any comorbidities Patients oral hygiene-related details included their practices of brushing and mouthwash use and frequency per day, use of fluoride toothpaste, any instructions received related to oral hygiene maintenance after treatment completion by the consultant, and the current use of dental prostheses.
Oral clinical parameters such as Oral Hygiene status was measured using Oral Hygiene Index – simplified (OHI-S) and categorized as good (0–1.2), fair (1.3- 3.0) and poor (3.1–6.0)27. The Oral Mucositis was assessed using WHO- Oral mucositis scale, and categorized into mild (by combining grade 1 and 2) and moderate to severe (by combining grade 3 and 4) categories28.
Outcome variable
OHRQoL is characterized as a self-report measure that focuses specifically on the oral health. it encompasses a comprehensive assessment of how oral diseases and conditions impact various aspects of an individual’s life. It assess the functionality as well as social and psychological effects stemming from oral health issues, aiming to understand their overall impact on well-being21. For this study we used Urdu version of European Organization of Treatment of Cancer QoL Head and Neck-35 (EORTC QLQ-H&N-35), which has been validated for use in our setting24.
EORTC QLQ-H&N-35 consists 35 questions in total, with 7 multiple item- questions assessing pain and soreness in the mouth and throat (PA) through item 31 -34, swallowing of liquid, pureed and solid food (SW) through item 35–38, senses of taste and smell (SE) from 43–44, social eating with family and friends and enjoying of meal (SO) 49–52 social contact with family and friends (SC) 48, 55–58 and sexuality (SX) 59–60 over the last week and 11 single questions to assess problems with “teeth” “opening of mouth” “dry mouth” ” sticky saliva” “coughing” “feeling ill” “ use of pain killers” “nutritional supplements” or “feeding tube” “weight loss” and “weight gain”. Likert type was used to score the responses as (1 = not at all, 2 = a little, 3 = quite a bit, 4 = very much) item H&N31 – H&N-35 have a 1 = yes, 2 = no response29.
Scoring of tool
First raw scores were calculated. For each multi- item question such as PA, SW, SO etc. an average of the corresponding items and for single- item question (teeth, mouth opening, sticky saliva, dry mouth etc.) the single score of the concerning item was taken as the raw score. Raw score = (I1 + I2 + I3)/n.
Standardization of the raw score was done to obtain mean scores for OHRQoL ranging from 0 to 100. Symptom scale S = [(RS-1)/range] × 100.
Elevated symptom scores for different domains of the tool implied deteriorating oral symptoms contributing to overall high mean score indicating more adversely affected OHRQoL.
Data collection procedure
Hiring of data collector was done prior to data collection. The training of data collector included procedure of taking informed consent, familiarization with the questionnaire and time management. A manual of operations was provided to data collector to facilitate consistency in protocol implementation during data collection. A trained dental hygienist was recruited to perform oral examination of the participants after they were interviewed.
Ethical clearance
An ethical review was sought from the Departmental Review Board (DRC), after its approval, the Ethical Review Committee (ERC) of Aga Khan University, Karachi, was consulted for ethical approval. The ERC approved the study (reference number 2022-7178-21416).
Statistical analysis
OHRQoL, which is the outcome variable is continuous in nature and calculated on the basis of a mean score. Multiple linear regression analysis was performed using STATA version 16.
The descriptive statistics of all independent variables and outcome variable was conducted. For categorical variables such as age, gender, level of education, monthly income, oral hygiene practices and other variables, frequencies and percentages were reported.
Normality of the outcome i.e. OHRQoL was checked using normal probability plot, which showed normal distribution. At the univariate analysis stage, all the categorical variables i.e. age, gender, marital status, BMI, educational status, employment status, monthly income, tumor stage, treatment type, radiation dose, number of follow up, smoking status and frequency, brushing and mouth wash use and their frequency, use of fluoride tooth paste, guidance related to oral complications of cancer treatment, visit to dentist, use of dental prosthesis were individually assessed for a significant association with OHRQoL on a set P value of ≤ 0.25.Variables with p-value greater, were removed and other variables with significant p-values were included in multivariable analysis. In Multivariable analysis, all variables that were significant at univariate analysis level, were assessed using multiple linear regression analysis. All variables with a p-value ≤ 0.05 were added in the model. The presence of biologically plausible interaction and confounding was also assessed.
After finalization of the main effect model, adequacy was assessed using residual plots against fitted values and normal probability plot.
Data management
Data was collected by a web-based structured questionnaire on google forms, which included mandatory questions, thereby, eliminating the issue of missing or incomplete data. The data was assessed for accuracy and completeness on daily basis by the investigator. Built-in range checks and internal consistency checks in google forms further ensured data quality. Backup files were created for data security, with participant information (such as name and contact details) excluded to maintain confidentiality. Each participant was assigned a unique ID number for data organization. The electronic data has been password- protected and will be deleted after seven years as per the policy of Aga Khan University.
Ethics approval and consent to participate
Ethical approval was obtained from Ethical Review Committee of the Aga Khan University Hospital, Karachi, Pakistan (reference number 2022–7178-21,416). Written informed consent was obtained from all the study participants. All methods were performed in accordance with the Declaration of Helsinki.
link

