Case study: facilitating bioethical goals using Blockchain technology
Applying BrainSwarming and the GPT to our case study, we generated 100 solution paths representing potential uses of blockchain technologies to further ethical objectives in clinical and research contexts (Fig. 2, Extended Data Table 1), thereby demonstrating the efficacy of the application of these techniques to the novel context of conceptual goals and intangible resources. For reasons of space, we highlight 25 of these potential solutions alongside their respective BrainSwarm solution pathways in Table 4 and provide more detailed explanations for five of these below. For ease of analysis and presentation, we further classified our solution pathways into ‘use concepts’—groups of solutions falling into thematic groups (Table 5). Several solutions were reached via multiple routes on the BrainSwarming graph—demonstrating the potential of these particular use cases to satisfy more than one bioethical goal.
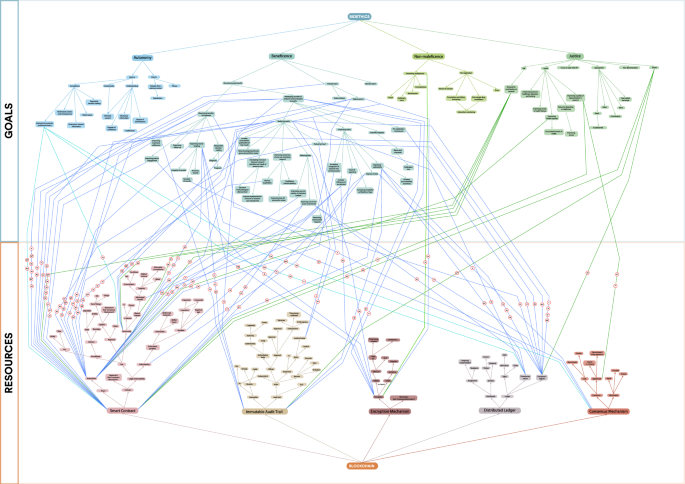
Application of Innovation Enhancing Techniques to a problem involving intangible goals and resources. BrainSwarm demonstrating 100 possible use cases of Blockchain technology to further ethical goals in healthcare and research. Numbering relates to possible use cases, expanded in Extended Data Table 1. * indicates the use case is highlighted and expanded in Table 4.
Examples of solution pathways
Below we outline five separate and distinct examples of solution pathways identified during our BrainSwarming process. We present these solution pathways as evidence that the application of IETs to contexts involving conceptual goals and intangible resources is feasible, and can indeed lead to effective identification of potential solutions to the specified problem. As with any other method in innovation, solutions identified by BrainSwarming, which pertains principally to the idea generation stage of the innovation process, must still be subsequently and rigorously evaluated according to their own merits before ultimately being implemented. We briefly explain each of the five example solution pathways below, refraining from detailed normative, technical, economic, or political assessment of the kind that would be necessary to complete the process of developing and implementing an innovation.
1. Conditional informed consent
Informed consent is a cornerstone of everyday clinical and research practice. However, it is often seen as a burden, overlooked, or implemented in ways that might shield a research project from legal liability but do little to respect the ideal of fully informed consent. In particular, many current consent procedures are largely static (patients may always withdraw from studies but otherwise their preferences cannot easily be updated) and unconditional (patients either consent or they do not).
Smart contracts are programs that execute on data contained in blockchains when specific conditions are met (e.g. ‘transfer payment sum if and only if title deeds are uploaded to property register’). If connected to a data oracle such that the relevant information is available to a blockchain-based smart contract, consents stored on a blockchain could be made conditional by automatically revoking consent should certain conditions arise. For example, consent for data use might be given for a fixed period or for certain uses only, or by certain individuals or groups. In addition to automaticity, a blockchain-based implementation of this use case would be transparent and tamper-proof.
2. Blockchain managed advanced directives
Advanced directives, also known as living wills, are documents expressing a person’s preferences towards future medical treatment and research participation decisions in the event of cognitive or other incapacity. Advanced directives suffer from several practical problems, including difficulty in accessing, verifying, and dating them, as well as the issues identified above relating to consent in general, such as being hard to update, unconditional, or insufficiently detailed or specific. Though issues relating to verification and dating can be addressed using professional services such as notarisation, these can be costly and time consuming. Dating advanced directives is of great importance, in part to assess whether they were made during a period of capacity but also because later versions of advanced directives are supposed to override earlier ones.
A possible solution to these issues, identified by our BrainSwarm, is to submit hashes of advanced directives to a blockchain-based registry. This has the potential to address issues of cost and effort (due to automation and removing the need for a notary, other witness, or lawyer), access (hashes of, but not the advanced directives themselves, would be publicly available on-chain), and ascertaining time (through the timestamping function of blockchains).
3. Tokenized incentives for treatment adherence/healthy behaviors
One of the most effective means of improving treatment adherence and healthy behaviour is through cash incentives13,14. However, these programs are not widely used in part due to ethical concerns over fairness, trade-offs, and opportunity costs12,15.
A potentially novel instantiation of this idea would be to reward treatment adherence and healthy behaviours with cryptocurrency tokens. A government could issue such tokens directly to individuals or through an intermediary and could imbue them with value by allowing them to be used, for example, for tax payments or for other government fees. Such a scheme would have the potential to reap the benefits of cash payments while obviating some of the associated ethical concerns, notably surrounding trade-offs and opportunity costs, as tokens would be free to mint and would not detract from other state health expenditures. The scheme could be set up to be financially self-sustaining by ensuring that payments made are outweighed by the overall money saved through improved population health.
4. Smart contract-based checklists for clinical trials
A significant proportion of biomedical research suffers from methodological flaws and a lack of statistical power16. Institutional and ethical review boards are supposed to review scientific merit in addition to legal compliance and ethical acceptability17 and are well-positioned to do so, since most human subject research has to undergo such an ethical review process. In practice, there is significant variability in the extent to which review boards attempt and are successful at ensuring scientific merit16,18.
Checklists have long been used to increase consistency and reduce errors in safety–critical contexts such as aviation and are increasingly applied in medicine and surgery19. A potentially novel application of this identified in our BrainSwarm is a smart contract-based checklist for institutional and ethical review. IRB members would fill out a review checklist on a hypothetical web portal. Progression through stages of protocol review would be locked by smart contract and predicated on submission of each section of the template. Upon submission, responses would be encrypted and sent to a repository via smart contract, which would also timestamp the submission. These responses could then be subject to random or automated audits.
The automated nature would alleviate potential concerns of ‘audit creep’ as it would not involve additional labour for IRBs. Timestamping could be used to document review process steps being taken in reasonable timeframes and in the correct sequence—not only providing incontrovertible evidence in cases of discrepancies or disputes, but also likely increasing the transparency of and trust placed in IRB review processes. Information on thoroughness and speed of reviews could also be used internally for quality improvement.
While the progress lock may be configured to force an IRB to make some kind of statement about the methodological merits or otherwise of proposed research, it is not intended to obviate or replace current protocols and legal frameworks, but rather to augment their implementation. Given the fundamental importance of basic scientific merit checks for overall scientific progress, however, any innovation which leads to improvements in this process would be worth weighing against these concerns. Other methods to develop the quality of proposals prior to submission should also continue to be utilised.
5. Ethical approvals released with published studies
Institutional and ethical review board decisions demonstrate a large degree of variability in the interpretation of regulations, value judgments, level of review required (full, none, or expedited), time to reach a decision, and quality of reasoning between different review boards17,20,21. Increasing transparency and accountability of ethical and institutional review, for example via publishing IRB decisions, has been proposed as a means of addressing these issues22. Such calls are sometimes resisted on the basis that increased transparency would be expensive and risks making public confidential information23.
A potentially novel means of addressing these concerns would be possible if ethics and institutional review are implemented on-chain, as outlined above. A smart contract could monitor trial publications and automatically decrypt and selectively publish review decisions relating to successfully published trials. The automated nature of this process would address expense concerns, while conditioning release of reviews on successful publication would partially address confidentiality concerns (as much of the potentially confidential information would be published anyway in the associated research paper) as well as contributing to transparency.
While separate confidentiality concerns related to IRB meeting minutes, memos, and other internal documents (rather than research protocols or participant data) are not addressed by this proposal, the fundamental role of the ethical review process in facilitating or inhibiting scientific progress makes any improvement in process, however partial, equally fundamental. These separate concerns could be addressed by having a specific form of review intended for publication alongside successful projects, which includes key information on ethics and methods reasoning but not more IRB-specific information such as meeting notes.
link

