May 26, 2023
Researchers at Mayo Clinic Comprehensive Cancer Center have found that deep learning technology used to automate segmentation for patients with head and neck cancer can save time without impacting contour accuracy. Results of the blinded, randomized trial were published in Frontiers in Oncology in April 2023.
The research team notes that head and neck cancer accounted for an estimated 5% of cancer-related mortality globally in 2020. Low- and middle-income countries are particularly impacted due to challenges in access to radiation therapy, chemotherapy and surgery. Radiation therapy is indicated for 74% of patients with head and neck cancer, making it critical in each patient’s care plan.
“Head and neck radiation treatment planning is resource-intensive and cumbersome,” says Samir Patel, M.D., a radiation oncologist at Mayo Clinic in Phoenix, Arizona. “This auto-contouring algorithm saved time in the cases we analyzed.”
Using Mayo Clinic’s comprehensive set of contouring guidelines for organs-at-risk (OARs), the research team trained a deep learning model and evaluated its feasibility as a reliable tool to reduce contouring time and increase contouring consistency.
Training the deep learning model
The research team retrospectively reviewed chart data to select 490 patient data sets to develop and evaluate the model. The researchers retrospectively re-contoured all the OARs for each of the cases, and were able to spend 11.6 hours per case to create a set of OAR contours for each of the data sets, creating a large and consistent data set for training.
Using this data set, the deep learning model was trained to automatically segment the OARs based on the planning CT (no MR or PET imaging was used). The model architecture is unique for this task — it is a 3D U-Net operated on a sliding window of 36 CT slices.
“We felt that this model struck a good balance,” says Joseph J. Lucido, III, Ph.D., medical physicist at Mayo Clinic in Rochester, Minnesota. “It has enough depth to handle the complexity of the contouring and is computationally tractable for model training and inference.”
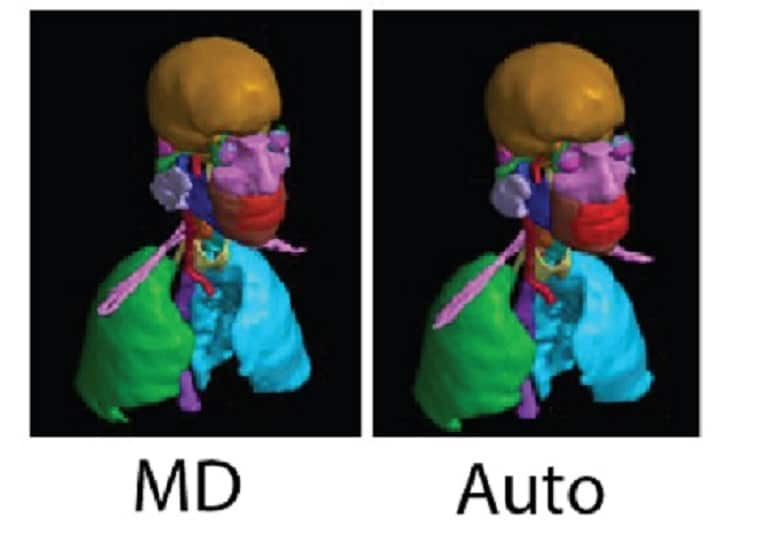
Autosegmented contours
This video shows a 3D view of the autosegmented contours.
Evaluation shows efficiency and accuracy
The study ran from December 6, 2021, through January 31, 2022, and involved eight radiation oncologists and eight medical dosimetry assistants. Twenty patient data sets were selected, balancing the location of the primary disease, surgical status and demographics.
For each data set, a medical dosimetry assistant created one set of OAR contours and the deep learning model created a different set. Radiation oncologists were blinded to the origin of the contours before reviewing and revising each set until they were acceptable for clinical treatment planning. To avoid recall, no radiation oncologist was assigned to both arms of the same patient data set. Each participating radiation oncologist reviewed an equal number of cases for each arm. One data set was excluded due to unintentional crossover in the blinding process, leaving 19 available for evaluation.
For every data set, the revision time required for the contours using the deep learning model was less than the combined time spent creating contours by the medical dosimetry assistant and the radiation oncologist’s subsequent review (MDA+RO time). The mean MDA+RO time was 3.4 hours, while the review time required using deep learning was .7 hours. This represents a time savings of 76%.
“The time savings associated with using this model allow our teams to help more people with head and neck cancer,” says Dr. Patel. “With further testing, this also may open up opportunities to address disparities in access around the world.”
Although nonsignificant statistically, radiation oncologist revisions also took 35% less time for the contours created by the deep learning model when compared with those created by the MDA.
Agreement with the gold standard was also equal to or significantly greater in the deep learning dataset when compared with the those created by MDAs and revised by radiation oncologists.
This initiative is the first research collaboration in a broader initiative between Mayo Clinic and Google Health, which aims to improve the delivery of care for serious and complex conditions. This strategic partnership harnesses knowledge and data to create digital tools available on innovative health care platforms that may reach patients worldwide. The next phase of this research will evaluate its use outside of Mayo Clinic.
For more information
Lucido J, et al. Validation of clinical acceptability of deep-learning-based automated segmentation of organs-at-risk for head-and-neck radiotherapy treatment planning. Frontiers in Oncology. 2023;13.
Refer a patient to Mayo Clinic.
link
